|
3/29/2024 0 Comments Density of water in gml
The density of the substance can be calculated as Ρ = 45 * 0.001 / 3.8 10 -5 = 1184 Įxample 2: Using Density to Identify a MaterialĪn unknown liquid substance has a mass of 18.5 g and occupies a volume of 23.4 ml (milliliter). The density of the golf ball can then be calculated as The volume of the golf ball can be calculated as Specific Weight for Some common MaterialsĪ golf ball has a diameter of 42 mm and a mass of 45 g. Specific Weight (or force per unit volume) can be expressed asĪ g = acceleration of gravity (9.807, 32.174 under normal conditions) Specific Weight is defined as weight per unit volume. See also Thermophysical Properties of Air - density, viscosity, critical temperature and pressure, triple point, enthalpi and entropi, thermal conductivity and diffusicity. Molecular weights can be used to calculate Specific Gravity if the densities of the gas and the air are evaluated at the same pressure and temperature. Ρ air = density of air (normally at NTP - 1.204 ) The Specific Gravity can be calculated as Specific Gravity of gases is normally calculated with reference to air - and defined as the ratio of the density of the gas to the density of the air - at a specified temperature and pressure. Specific Gravity for some common Materials Specific Gravities common Substances Substance See also Thermophysical Properties of Water - Density, Freezing temperature, Boiling temperature, Latent heat of melting, Latent heat of evaporation, Critical temperature. Water is normally also used as reference when calculating the specific gravity for solids. 
SG of a fluid has the same numerical value as its density expressed in g/mL or Mg/m 3. Since Specific Gravity - SG - is dimensionless, it has the same value in the SI system and the imperial English system (BG). It is common to use the density of water at 4 oC (39 oF) as a reference since water at this point has its highest density of 1000 kg/m 3 or 1.940 slugs/ft 3. 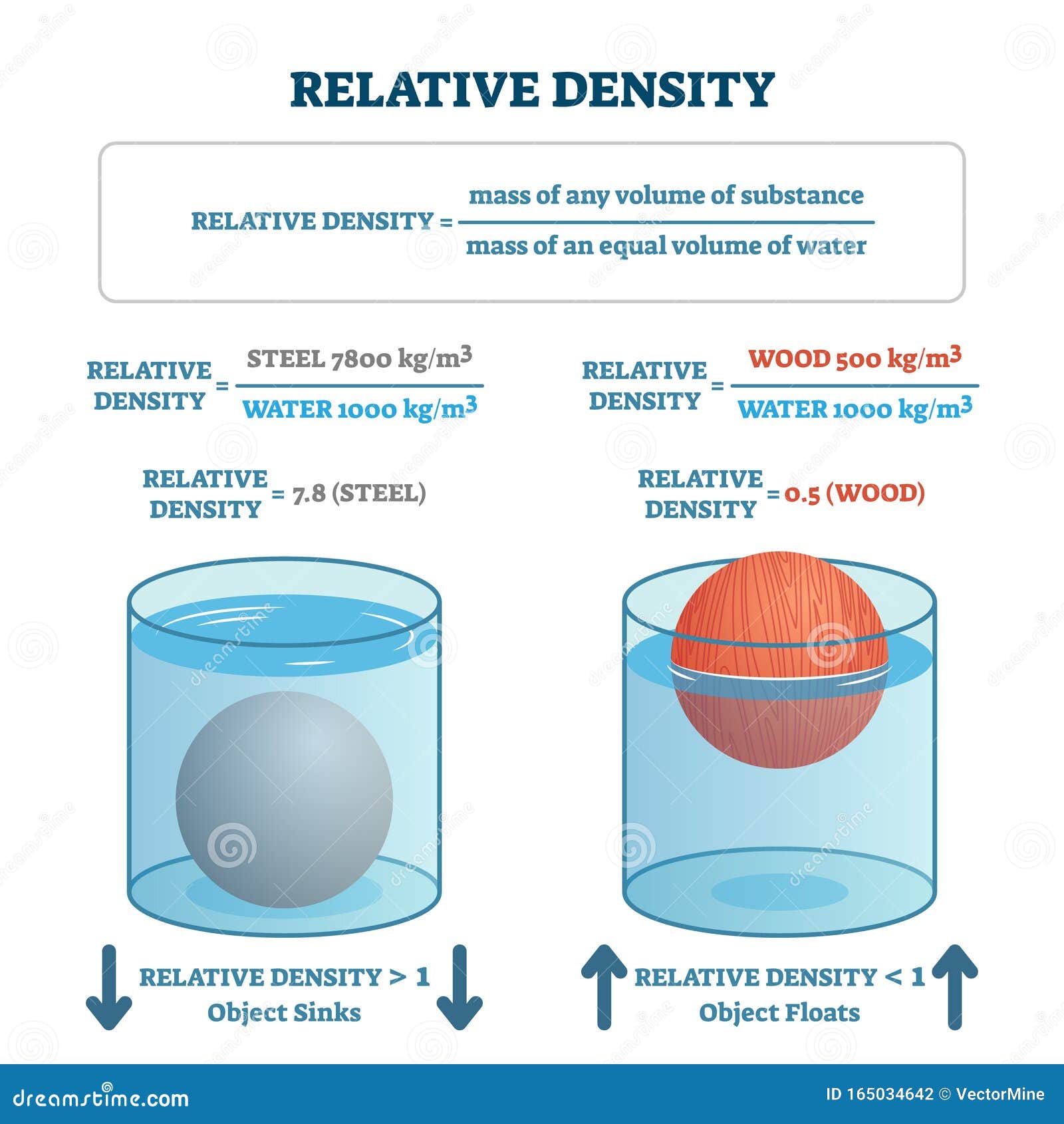
Ρ H2O = density of water - normally at temperature 4 oC Ρ substance = density of the fluid or substance Specific Gravity (Relative Density) - SG - is a dimensionless unit defined as the ratio of the density of a substance to the density of water - at a specified temperature and can be expressed as How to measure density of liquid petroleum products.Air - Density, Specific Weight and Thermal Expantion Coefficient - variation with temperature and pressure, SI and Imperial units.Water - Density, Specific Weight and Thermal Expantion Coefficient - variation with temperature at 1, 68 and 680 atm, SI and Imperial units.Density is a physical property - constant at a given temperature and pressure - and may be helpful for identification of substances.īelow on this page: Specific gravity (relative density), Specific gravity for gases, Specific weight, Calculation examples On atomic level - particles are packed tighter inside a substance with higher density. See also Unit converter - mass and Unit converter - density Slugs can be multiplied with 32.2 for a rough value in pound-mass (lb m). Note that there is a difference between pound-force (lb f) and pound-mass (lb m). The Imperial (U.S.) units for density are slugs /ft 3 but pound-mass per cubic foot - lb m /ft 3 - is often used. What is weight and what is mass ? - the difference between weight and mass.Mass is a property and the SI unit for density is. Similarly, a decrease in solute concentration decreases the specific gravity of urine, which indicates medical problems like renal failure.Density is defined as mass per unit volume. An increase in the specific gravity of urine indicates that it is due to an increase in the solutes caused by dehydration, diarrhea, or infection. Both high and low density or specific gravity than the normal range of urine indicates medical problems. Both the blood and urine have dissolved substances in water that increase the density from that of pure water. The density of urine varies in the range of 1.0050 g/mL to 1.030 g/mL. For example, the density of whole blood for humans is ~1.060 g/mL. When substances dissolve in water, the density of the solution is usually different from pure water. The density of water is 1.0 g/mL at room temperature, so the specific gravity is equal to the density of the object expressed without a unit. Therefore, the specific gravity is a unitless number.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
